Clinical Revelance
- Demonstrates high analytical sensitivity, with the ability to reliably and consistently detect few HPV 16 or HPV 18 DNA copies present.
- Exhibits quantitative detection of HPV across a broad dynamic range, enabling high-resolution molecular monitoring for superior correlation with therapeutic benefit.
- Displays low quantitative variability (<30% CV above 5 copies).
- Demonstrates low level of background signal (<0.04 copies per sample across 20 healthy donor samples), indicating high specificity.
Treatment response
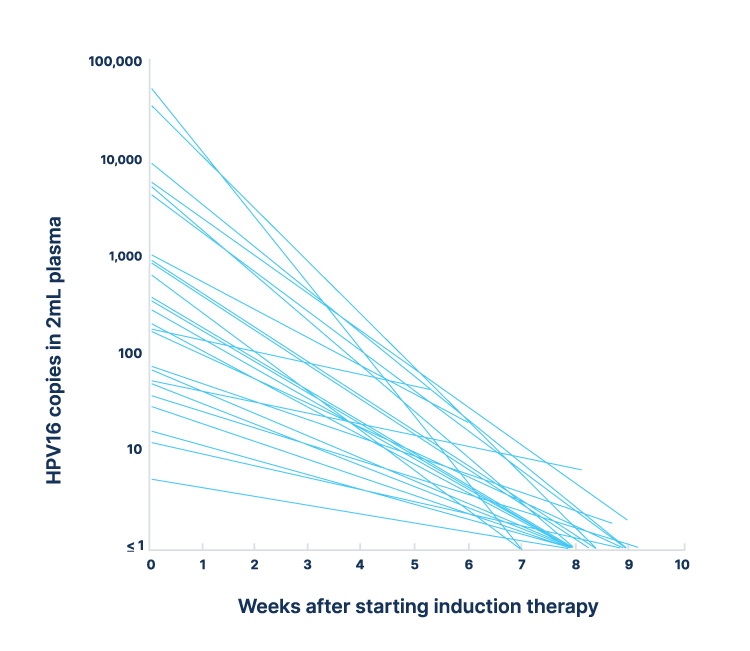
HPV-SEQ has been employed to evaluate dynamic changes in circulating HPV alongside patients’ radiographic assessment of therapy response to assess future utility in guiding treatment de-escalation strategies.
Disease surveillance and beyond
Demonstrating ultra-sensitive detection of cfHPV-DNA, HPV-SEQ may be used during and at conclusion of cancer treatment to assess the diminishing presence or total absence of cfHPV-DNA as a surrogate of disease burden.